Boehringer Ingelheim refines its bioprocess technology operations in Animal Health with greater focus on environmental sustainability to reduce footprint
Press Release
From replacing plastic roller bottles to the adoption of chemically defined serum-free substances to allow efficient cell culture. From optimizing process chains for antigenic biologicals manufacturing to continuously discussing and evaluating current practices, Boehringer Ingelheim is strongly committed to reducing its environmental footprint. Since biotechnology revolutionized the pharma industry in the 1980s by safely producing medicines derived from living cells, scientists have been working on solutions to make these processes more sustainable. That is the goal of our Bioprocess Technology Shift, a global program in charge of integrating innovative technologies and processes into Boehringer Ingelheim’s Animal Health vaccine manufacturing operations.
The program was launched in 2017 in order to prepare biotechnological manufacturing operations for the future. It brings together expertise from two vital areas: Operations and Research & Development. Bernd Eichenmüller, Global Head of the Bioprocess Technology Shift Program, works closely with Noël Detraz, Global Head of Bioprocess Development and Industrialization (BPDI).
“The overarching idea is to produce smaller and fewer batches with defined ingredients and controlled processes – meaning less water consumption, smaller footprint, less waste. It is critical that the development of these processes starts in R&D and transitions smoothly into manufacturing, hence our close collaboration. On the streets there is the Fridays for future movement, and in our labs, every day is for future,” explains Noël.
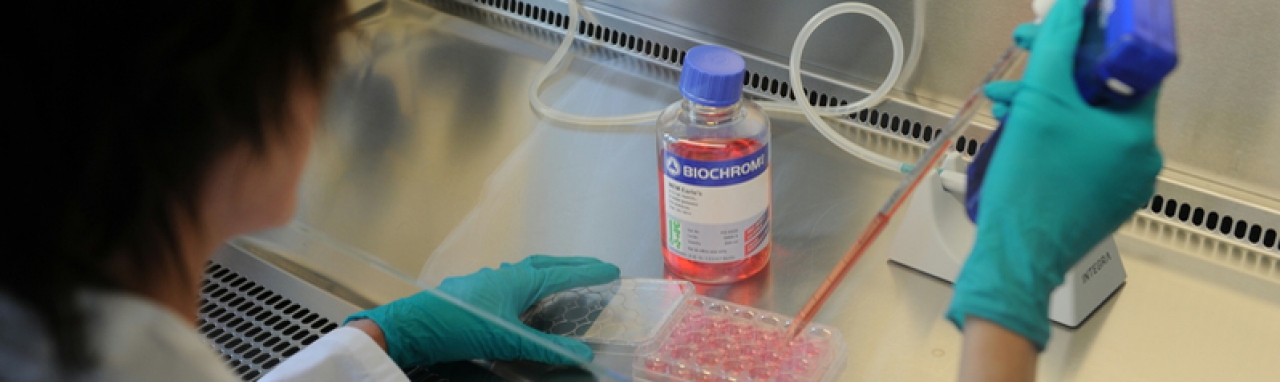
Upstream cell culture in laboratory for Animal Health products
Social responsibility is a key driver of this initiative. Consequently, Boehringer Ingelheim wants to change, standardize processes and reduce complexity in manufacturing by using platform technologies and equipment in all manufacturing sites across the world to make them more environmentally friendly. A good example are the efforts to say goodbye to roller bottles.
“Roller bottles are plastic. Furthermore, they need to be manually opened several times during a production process, something that is labor intensive and greatly increases the risk of contamination. Instead, we are switching to bioreactors that are between 1,000 and 10,000 times larger, in a process that is more controlled, robust and efficient,” says Bernd.
Reducing waste and water consumption is also a great concern. That is already possible by utilizing single-use technology which does not require extensive sterilization with energy consuming steam or cleaning with large amount of water and cleaning solutions. In addition, the development of processes that yield higher amounts of product per batch ultimately means fewer batches, less cleaning, less water usage.
Having a greener production means even more. Boehringer Ingelheim is also shifting away from animal-origin and high-risk materials, like animal-derived serum, which traditionally has provided the needed nutrients to enable cell growth. They pose a certain amount of risk, as they may contain extraneous agents like viruses, and often cannot be sustainably supplied.
“Nowadays, we can apply serum-free conditions to cell cultures using fully characterized media and completely avoid this risk,” explains Bernd. “When you combine the efforts of closed operations and defined media, you can even get rid of antibiotics that have been used in the past.”
Noël adds another benefit of this program: “Technology has advanced so much that in some cases new lab tests can reduce assay variation and provide more batch-to-batch consistency. There are good reasons for moving in this direction”.
Is it possible to reach a 100% sustainable production process? The answer is yes, but some patience and time are still needed. When dealing with biologicals, it can take many years due to regulatory requirements and approvals for process and/or material changes.
“Change is not immediate in all cases, but our improvement is. Our team sees short and midterm gains, including achieving a significant impact through higher yields, improved process robustness, lower cost of goods, higher quality, improved safety and animal welfare, and faster time to market,” observes Bernd.
Article and Image Source: